About Magnesium Sulfate
Magnesium sulfate (MgSO4) is the safest, most effective, and lowest-cost medication to treat life-threatening convulsions among women during pregnancy and childbirth.
Each year approximately 287,000 women die from pregnancy and childbirth-related complications. Pre-eclampsia and eclampsia (PE/E) – the dangerous elevation of blood pressure during pregnancy – is the second leading cause of maternal death and can lead to seizures, kidney and liver damage, and both maternal and infant deaths if untreated. It is attributed to 22% of all maternal deaths, affecting an estimated 63,000 women worldwide each year[1].
Commodity Introduction
Magnesium sulfate (MgSO4) is recognized by the WHO as the safest, most effective, and lowest-cost medication for treating PE/E. Magnesium sulfate is the standard treatment for these conditions in the majority of developed countries, but less-effective and riskier medications, such as diazepam and phenytoin, still are widely used in developing countries.
Magnesium sulfate costs approximately US $0.10 per ml (supplier median price), and is produced by a number of global manufacturers and many local manufacturers worldwide. Magnesium sulfate is administered by injection into the woman’s vein or muscle. Calcium gluconate—a mineral supplement—is an antidote available in the rare event of magnesium sulfate overdose.
Commodity Context
Magnesium sulfate has been placed on the World Health Organization (WHO) Essential Medicines List (EML) and is recommended as the most effective, safe and affordable treatment “for use in eclampsia and severe pre-eclampsia and not for other [seizure] disorders[2].”
Case studies show magnesium sulfate is included in national protocols for maternal health service provision as well as EMLs and standard treatment guidelines in many countries (where national data was collected and reported), including Ghana, Mali, Malawi, Mongolia, and India[3]. Furthermore, a study by MCHIP states that most countries—except Rwanda and Ethiopia—reported that they have magnesium sulfate on their EMLs[4]. A WHO survey related to the Global Reproductive Health Strategy (World Health Assembly Resolution 55.12) assessed the implementation of magnesium sulfate and found that in about 85% of the surveyed 58 countries, magnesium sulfate was available for use.
Products Available
Around 35 manufacturers of generic magnesium sulfate solutions have been identified worldwide but there are likely to be other manufacturers around the world, especially in Asia and Latin America. Products are available in glass ampoules or vials sealed with a rubber or latex stopper and cap. The product is very stable at ambient temperatures and is unlikely to undergo any significant degradation as a result of heat if it is properly manufactured, packaged, sterilized and sealed. Recent consultations among experts have led to the recommendation of one particular dosing: 5g in 10mL.
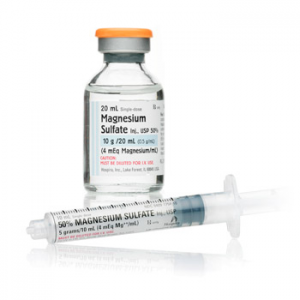
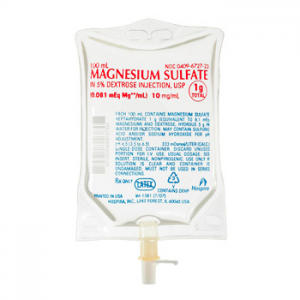
Source: Hospira
Product Efficacy
Large clinical trials conducted between 1995 and 2002 found magnesium sulfate to be the most effective when compared with other treatments, including diazepam and phenytoin[5]. These trials have found magnesium sulfate to reduce the occurrence of eclampsia by more than 50% and maternal deaths by 46%. Women treated with magnesium sulfate had a 52% and 67% lower recurrence of convulsions (eclampsia) than those treated with diazepam and phenytoin, respectively.
Common Barriers
Barriers to the effective use of magnesium sulfate in developing countries include complex dosing regimens, a lack of product harmonization, and low provider familiarity of the drug, all of which limit its impact[6].
For example, a survey conducted by WHO revealed the following challenges: providers’ continued reliance on diazepam due to relative ease of use in some rural facilities; hesitancy of providers to administer magnesium sulfate due to perceived side effects; and delays in getting the second dose when health centers transfer the patient to upper level of care after giving the first dose. In some cases, magnesium sulfate use was limited to specialized care facilities. National guidelines detailing obstetric roles for intermediate facilities and training health providers in administering magnesium sulfate could improve the appropriate use of this medicine[7].
For more information on Magnesium Sulfate, please visit the UN Every Woman Every Child initiative webpage.
[1] EngenderHealth. (2007). Balancing the Scales: Expanding Treatment for Pregnant Women with Life-Threatening Hypertensive Conditions in Developing Countries;sx A Report on Barriers and Solutions to Treat Pre-eclampsia & Eclampsia.
[2] Organization, World Health. (2013). WHO model list of essential medicines: 18th list (updated) April 2013.
[3] PATH, MSH, UNFPA, USAID. (2012). Medicines for Maternal Health: United Nations Commission on Life-Saving Commodities for Women and Children.
[4] Fujioka A, Smith J. (2011). Prevention and Management of Postpartum Hemorrhage and Pre-Eclampsia/Eclampsia: National Programs in Selected USAID Program-Supported Countries.: Maternal and Child Health Integrated Program (MCHIP).
[5] Altman, D, Carroli, G, Duley, L, Farrell, B, Moodley, J, Neilson, J, & Smith, D. (2002). Do women with pre-eclampsia, and their babies, benefit from magnesium sulphate? The Magpie Trial: a randomised placebo-controlled trial. Lancet, 359(9321), 1877-1890.
[6] PATH. (2013). Breakthrough innovations that can save women and children now. from http://www.path.org/innovations2015/
[7] PATH, MSH, UNFPA, USAID. (2012). Key Data and Findings: Medicines for Maternal Health.

 This website is made possible by the support of the American People through the
This website is made possible by the support of the American People through the